Neutralisation Reaction: Difference between revisions
No edit summary |
No edit summary |
||
| Line 8: | Line 8: | ||
When we talk about how acidic something is, we generally talk in terms of pH. pH exists as a 1 – 14 scale, with 7 being a perfectly neutral pH – this is where pure water sits. | When we talk about how acidic something is, we generally talk in terms of pH. pH exists as a 1 – 14 scale, with 7 being a perfectly neutral pH – this is where pure water sits. | ||
[[File:PH_scale.jpg|600px|center| | [[File:PH_scale.jpg|600px|center|pH scale]] | ||
Acidic and alkaline substances, when combined, will react to form a salt, with water as a ‘waste product’. The acid and alkali will neutralise each other, and bring the pH closer to the neutral 7. | Acidic and alkaline substances, when combined, will react to form a salt, with water as a ‘waste product’. The acid and alkali will neutralise each other, and bring the pH closer to the neutral 7. | ||
| Line 24: | Line 24: | ||
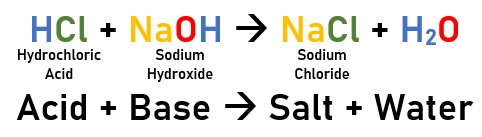
These two undergo the neutralisation reaction when mixed to form Sodium Chloride, i.e. table salt. | These two undergo the neutralisation reaction when mixed to form Sodium Chloride, i.e. table salt. | ||
[[File:NeutralisationReactionNaCl.jpg|center| | [[File:NeutralisationReactionNaCl.jpg|center|Neutralisation Reaction]] | ||
The pH of the mixture evens out to a neutral 7. | The pH of the mixture evens out to a neutral 7. | ||
Revision as of 14:18, 13 March 2023
Many mineral supplements are created by the Neutralisation Reaction.
What is the neutralisation reaction?
When we talk about how acidic something is, we generally talk in terms of pH. pH exists as a 1 – 14 scale, with 7 being a perfectly neutral pH – this is where pure water sits.
Acidic and alkaline substances, when combined, will react to form a salt, with water as a ‘waste product’. The acid and alkali will neutralise each other, and bring the pH closer to the neutral 7. If the correct amount of each is used, the resulting salt will have the perfect neutral pH of exactly 7.
Let's take table salt as an example.
Table salt, also known as Sodium Chloride (NaCl), is created by a neutralisation reaction, and generally sits at a pH of 7.
Hydrochloric acid (HCl) is very acidic, with a pH of about 3. Sodium Hydroxide (NaOH) is very alkaline, with a pH generally between 10 and 13.
These two undergo the neutralisation reaction when mixed to form Sodium Chloride, i.e. table salt.
The pH of the mixture evens out to a neutral 7.